Xellfree®
Cell-free Allografts
Allografts, wie z.B. menschliche Herzklappen, sind anatomisch perfekt an ihre Aufgabe angepasst. Daher zeigen Allografts, früher auch als "Homografts" bekannt, grundsätzlich gute Langzeitergebnisse. Der Nachteil von konventionellen Allografts ist, dass sie vom Immunsystem erkannt werden können. Dies kann zu einem Funktionsverlust des Allografts führen und eine Reintervention oder Reoperation erfordern.
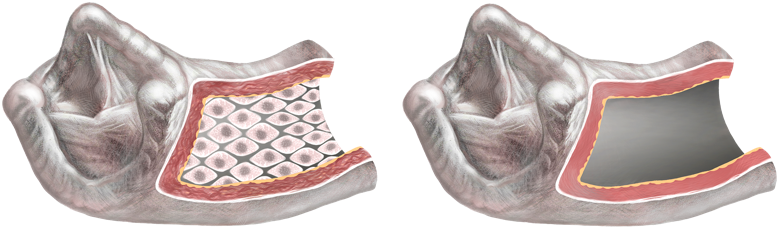
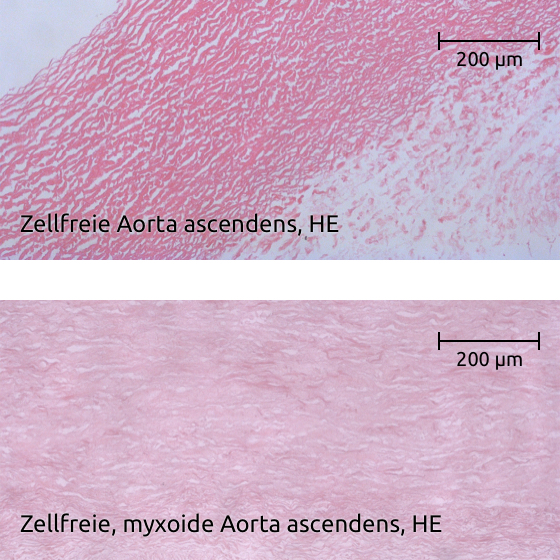
Bei der "Dezellularisierung" werden die Zellen aus der Gewebespende entfernt. Es bleibt nur das zellfreie Bindegewebe übrig. Das nun zellfreie Allograft behält seine ursprüngliche Form und volle Funktionsfähigkeit. Während Zellmarker die Individualität jedes einzelnen Menschen bestimmen, ist das Bindegewebe nicht immunologisch individualisiert. Zellfreie Allografts werden daher seltener als konventionelle (kryokonservierte) Allografts vom Empfänger abgestoßen.
Xellfree®Process
Discover more in this slideshow about the Xellfree®-process.
Clinical experience
Cell-free human heart valves are always implanted with a more or less long piece of the adjacent vessel (ascending aorta, truncus pulmonalis) that carries the valve.
No cellular immunogenicity
Neumann et al. (2014) untersuchten über drei Jahre das periphere Blut von 47 Patientinnen und Patienten, die zellfreie Pulmonalklappen erhalten haben. Es konnten keine erhöhten Spiegel an T-Zellen (CD3+, CD8+), B-Zellen (CD19+), Killer-Zellen (CD16+/CD56+) oder T-Helferzellen (CD4+) nachgewiesen werden. Die zellfreien Pulmonalklappen waren immunologisch stumm.
“Early Systemic Cellular Immune Response in Children and Young Adults Receiving Decellularized Fresh Allografts for Pulmonary Valve Replacement”
Cellular Restoration
Sarikouch et al (2019) investigated the postoperative immigration of cells into decellularized allograft. The relative histologic score (100% corresponds to complete cellular restoration) in samples that did not exhibit endocarditis (n=6) was 76±4%. Intracellular procollagen type 1 was found in the mesenchymal recipient cells. In endocarditis samples (n=5), the histological score was significantly lower at 48±7%, associated with leukocyte infiltration and matrix degradation. A cell-free pulmonary valve showed an immune system related graft failure. Allografts sampled in the first 12 months post-op showed a thin colonization of the media. In terms of the degree of restoration, no difference was found between a cell-free aortic or pulmonary heart valve.
“Early Insight Into In Vivo Recellularization of Cell-Free Allogenic Heart Valves”
Cell-free pulmonary valve
Bis 2014 waren jahrzehntelang Rinderhalsvenen (Bovine Jugularvene, BJV) und konventionelle, kryokonservierte Allografts (kryokonserviert) die verbreitetsten Optionen für den Pulmonalklappenersatz bei angeborenen Herzfehlern. Seit 2014 haben sich zellfreie, humane Pulmonalklappen, Espoir PV als Alternative etabliert. Bobylev et al. verglichen 2024 den klinischen Verlauf von 319 „propensity-matched“ Patientinnen und Patienten (Alter, Herzfehlertyp, Anzahl der vorherigen Eingriffe) nach Pulmonalklappenersatz mit einer BJV oder mit einer Espoir PV. Die Espoir-Klappen zeigten deutlich bessere 10-Jahres-Ergebnisse als BJV, mit statistisch signifikant besserer Freiheit von Endokarditis (97% vs. 87%), Explantation (96% vs. 82 %) und Degeneration (65% vs. 40%).
„Matched comparison of decellularized homografts and bovine jugular vein conduits for pulmonary valve replacement in congenital heart disease“
Im Jahr 2022 verglichen Bobylev et al. den klinischen Verlauf von 319 Patientinnen und Patienten (gleiche „Match“-Kriterien wie oben) nach Pulmonalklappenersatz mit einem konventionellen, kryokonservierten Allograft oder mit einer Espoir PV. Auch hier zeigte sich eine signifikant bessere Freiheit von Explantationen für die Espoir-Klappe im Vergleich zum kryokonservierten Allograft (96% vs. 83%) und bessere Freiheit von strukturellen Klappenveränderungen nach 10 Jahren (Espoir 65% und kryokonserviert 47%).
„5-Year results from the prospective European multi-centre study on decellularized homografts for pulmonary valve replacement ESPOIR Trial and ESPOIR Registry data“
Die positiven klinischen Daten bestätigen die Überlegenheit der zellfreien humanen Pulmonalklappen, Espoir PV gegenüber konventionellen Optionen – auch nach mehr als 10 Jahren post-OP.
Cell-free aortic valve
Bis 2015 galten die Implantation einer konventionellen Herzklappenprothese (Bioprothese oder mechanische Prothese) sowie die Ross-Operation als die gängigsten Verfahren für den Aortenklappenersatz. Seitdem haben sich die zellfreien humanen Aortenklappen, Arise AV, als vielversprechende Alternative etabliert. Anfang 2024 beschrieben Horke et al. den klinischen Verlauf von 358 Patienten (Medianalter 23,9 Jahre), von denen 144 im Rahmen der multizentrischen ARISE-Beobachtungsstudie (2015–2018) prospektiv erfasst wurden. Eine Subgruppe (n= 215, Medianalter 39,2 Jahre) wurde in einem Propensity-Matching-Verfahren (unter Berücksichtigung von Alter, Herzfehlertyp und Anzahl vorheriger Eingriffe) mit ähnlichen Kohorten verglichen, die konventionelle Prothesen erhielten oder eine Ross-Operation durchliefen. Die Ergebnisse zeigen, dass Arise-Klappen über einen Beobachtungszeitraum von Median 5 Jahren (max. 12 Jahre) weniger unerwünschte Nebenwirkungen aufwiesen als die Vergleichsgruppen.
„Five-year results from a prospective, single-arm European trial on decellularized allografts for aortic valve replacement-the ARISE Study and ARISE Registry Data“
Eine Subgruppenanalyse im Kontext des Aortenklappenersatzes ist klinisch relevant, da pädiatrische Patienten aufgrund ihres ausgeprägteren Immunsystems ein erhöhtes Risiko für Abstoßungsreaktionen aufweisen. Horke et al. untersuchten speziell diese Subgruppe (n = 143, Medianalter 10,8 Jahre). Die frühzeitige Mortalität nach Arise-Klappenimplantation war signifikant niedriger im Vergleich zur Ross-Operation (0,7 % vs. 3,7 %). Das mittelfristige Überleben lag bei 97,8 % nach fünf Jahren und bei 96,3 % nach zehn Jahren postoperativ. Unerwünschte Ereignisse wie Reoperationen, strukturelle Klappenveränderungen, Endokarditis oder thromboembolische Komplikationen traten in der Arise-Kohorte nach einer mittleren Nachbeobachtungszeit von 5,3 Jahren mit einer vergleichbaren Häufigkeit auf wie in der Ross-Kohorte. Allerdings hatten Patienten mit Arise-Klappenimplantation deutlich häufiger kardiale Voreingriffe (59 % vs. 32,3 %) und insbesondere häufiger Eingriffe an der Aortenklappe (17 % vs. 2,3 %).
Arise-Klappen sind eine therapeutische Option für pädiatrische Patienten mit eingeschränkter linksventrikulärer Funktion, da der Eingriff auf den Ersatz der betroffenen Klappe beschränkt bleibt und die Möglichkeit einer späteren Ross-Operation bewahrt wird. Die Wahl der geeigneten Operationsmethode erfordert letztendlich eine patientenindividuelle Beurteilung unter Berücksichtigung anatomischer und myokardialer Parameter sowie spezifischer Risikofaktoren für thromboembolische Ereignisse oder Blutungskomplikationen.
“Paediatric aortic valve replacement using decellularized allografts: a multicentre update following 143 implantations and five-year mean follow-up”
Replacing the aortic and pulmonary valve
Cell-free human heart valves are also suitable for double valve replacement. Since the cell-free design leads to less rejection, matching heart valves from different donors can be implanted (Bobylev 2019).
“Double semilunar valve replacement in complex congenital heart disease using decellularized homografts.”